Developing a High-Throughput CRISPR Screening Approach to Discover Genes Driving Human Tissue Injury Response
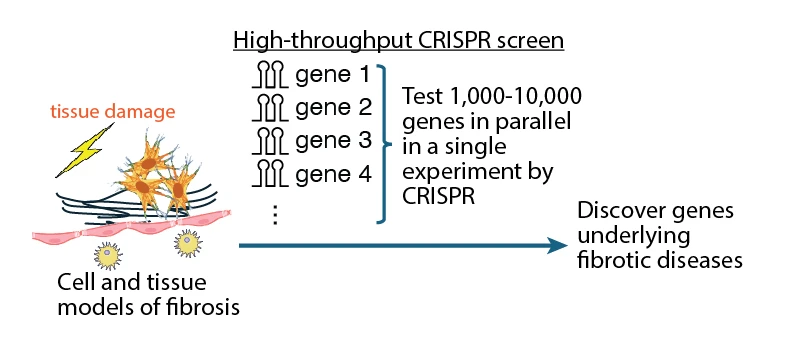
Dr. Chris Hsiung, an assistant professor at the University of California-San Francisco (UCSF), and his colleagues have developed new methods for high-throughput CRISPR screens, a technology that enables the perturbation of thousands to tens of thousands of genes or their combinations in a single compressed experiment to test their contributions to a given biological process. They are developing new ways to use these approaches to discover genes that may serve as therapeutic targets for diseases of high global burden.
A major challenge to understanding and treating common diseases is that it is often unknown which of the ~20,000 genes in the human genome contribute to such diseases and therefore can be targeted therapeutically. Moreover, common diseases often arise from the contributions of multiple genes. Conventional approaches that test genes one-by-one is laborious and ineffective. More scalable approaches for discovering combinations of genes as potential therapeutic targets are needed. Support from the Bachrach family is enabling Dr. Hsiung and his team to develop new strategies of using high-throughput CRISPR screening to discover therapeutic targets in fibrosis. Fibrotic diseases affect vital organs like the lungs, liver, kidneys, and heart, contributing to 18% of all global deaths. Fibrosis also contributes to the development of cancer, which accounts for an additional 17% of global deaths. This work holds promise to develop new treatments for a pathophysiologic tissue damage response that contributes to a large fraction of global morbidity and mortality.

