Testing the Effectiveness of a New Regenerative, Anti-Inflammatory Therapy for Chronic Pouchitis
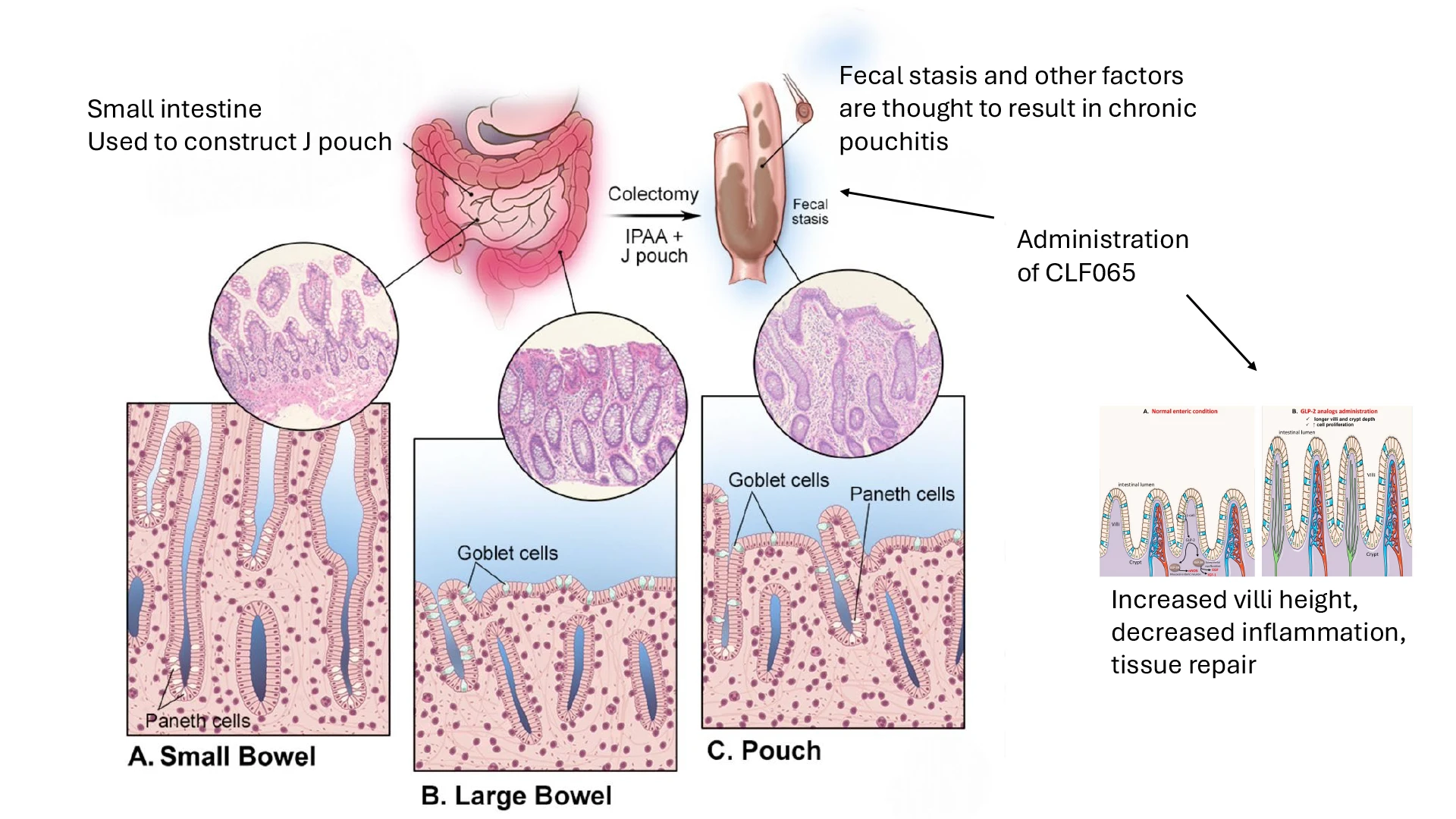
Dr. Amy Lightner at the Calibr-Skaggs Institute for Biomedical Innovation is leading a Phase 2 randomized, double blinded placebo controlled clinical trial to evaluate a long-acting GLP-2 analogue, CLF065, as a novel treatment for chronic pouchitis. The study aims to offer the first FDA-approved regenerative therapy for this debilitating condition.
Pouchitis is a painful and chronic inflammatory condition that affects nearly half of patients who undergo surgery for ulcerative colitis. Despite its prevalence, no FDA-approved therapies currently exist for chronic pouchitis, leaving patients dependent on long-term antibiotics or immunosuppressants with limited effectiveness. CLF065 is a long-acting analogue of glucagon-like peptide-2 (GLP-2), a naturally occurring hormone known to promote intestinal epithelial repair, enhance mucosal barrier function, and reduce inflammation. GLP-2 has been shown in both preclinical and clinical studies to increase villi height in the small intestine, reflecting enhanced mucosal surface area and nutrient absorption. In the setting of pouchitis, where epithelial integrity is compromised and patients experience symptoms like bowel frequency and urgency, increased villi height may contribute to improved absorptive capacity and reduced fluid loss—thus potentially alleviating key clinical symptoms. This randomized, placebo-controlled Phase 2 trial investigates whether CLF065 can safely and effectively treat chronic pouchitis using clinical, endoscopic, histologic, biomarker, and patient-reported outcome measures. Treatment success will be assessed through improved symptoms, decreased ulceration on endoscopy, and histologic evidence of mucosal healing, including increased villi height, a structural marker of improved intestinal health rather than a direct mediator of inflammation. If successful, this trial could not only transform the standard of care for chronic pouchitis but also illuminate novel regenerative mechanisms applicable to other forms of inflammatory bowel disease, laying the groundwork for regulatory approval of a new class of gut-targeted regenerative therapies.

